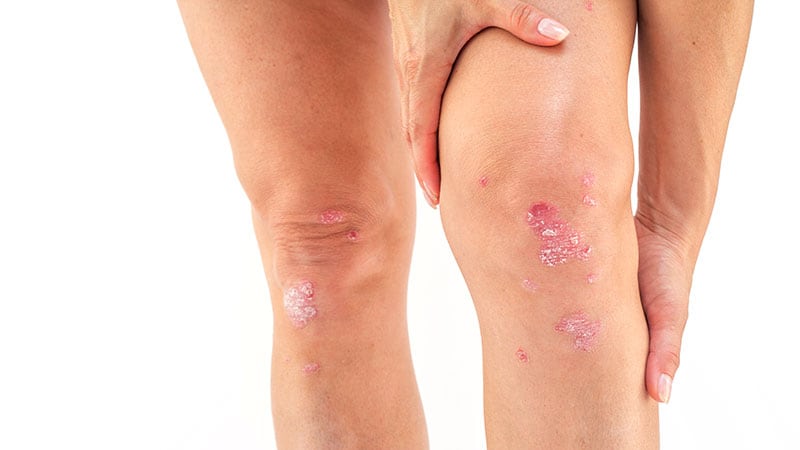
National Institute for Health and Care Excellence (NICE) has recommended guselkumab (Tremfya) as a treatment option for moderate to severe plaque psoriasis.
Guselkumab is recommended for the treatment of adults with plaque psoriasis if the following conditions are fulfilled:
- Severe disease: Psoriasis Area and Severity Index (PASI) of ≥10 and Dermatology Life Quality Index (DLQI) of >10.
- Inadequate response, intolerance or contraindication to other systemic therapies, including ciclosporin, methotrexate, and psoralen+long-wave ultraviolet A radiation.
The recommended dose for guselkumab is 100 mg given subcutaneously at weeks 0 and 4, and subsequently 100 mg maintenance dose every 8 weeks. Therapy should be discontinued if psoriasis has not responded adequately after 16 weeks. An adequate response is defined as a 75% reduction in PASI or a 50% reduction in PASI+5-point reduction in DLQI.
Evidence has shown guselkumab is more effective than TNF-alpha inhibitors (adalimumab, etanercept, and infliximab) and ustekinumab, whereas its health benefits may be comparable to ixekizumab and secukinumab. Its cost-effectiveness may be similar to or better than ixekizumab and secukinumab.
The drug is to be used only when provided by the company in agreement with the commercial arrangement. The recommendations should not affect patients whose treatment with the drug was started before publication of the guidance.