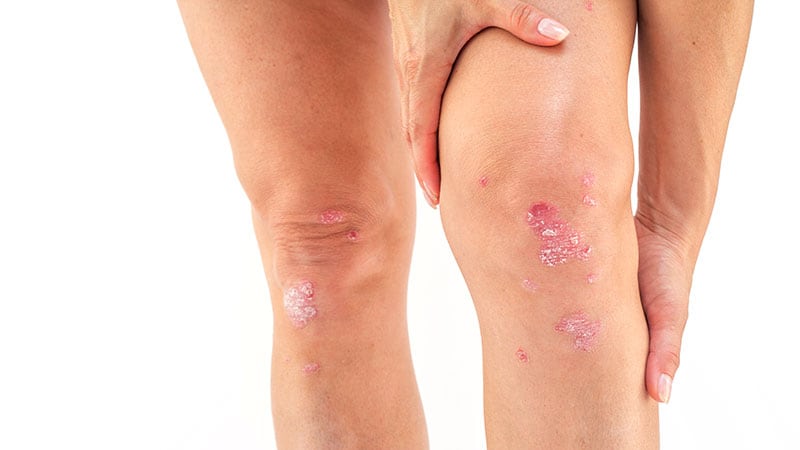
Takeaway
- Secukinumab is associated with clinical benefit in patients with moderate to severe plaque psoriasis, according to results from the real-world PROSPECT study.
Why this matters
- Cohort included prior and concurrent users of other systemic therapy.
Key results
- At week 24, secukinumab was associated with 86.1% of patients achieving ≥75% improvement in the Psoriasis Area and Severity Index (PASI75).
- 68.5% of patients achieved PASI90.
- 39.7% of patients achieved PASI100.
- In a subgroup analysis of patients naive to systemic therapies, 92.8% achieved PASI75, 84.3% achieved PASI90, and 66.3% achieved PASI100.
- In patients previously treated with biologics, 78.1% achieved PASI75, 54.8% achieved PASI90, and 29.0% achieved PASI100.
- 45.9% of patients experienced an adverse event.
- Most common were nasopharyngitis (8.7%), pruritus (2.9%), and headache (2.4%).
- 4.1% of patients experienced a serious adverse event.
- 6.9% of patients discontinued because of an adverse event.
Study design
- Efficacy data for 1323 of 1988 patients from an ongoing prospective study completing 24 weeks of treatment with secukinumab for moderate to severe plaque psoriasis.
- Of the overall cohort, 90.9% had received prior systemic therapy and 44.3% were receiving concomitant treatment.
- Funding: Novartis Pharma GmbH.
Limitations
- No comparator group.
- Short study length.
References
References