A combination of existing drugs may emerge as the best treatment for COVID-19 rather than the emergence of a single 'big winner', according to the leaders of a major UK trial.
The Randomised Evaluation of COVID-19 Therapy (RECOVERY) trial, led by the University of Oxford, is the world's largest randomised clinical trial of potential COVID-19 treatments.
Over the last 2 months it has recruited more than 10,000 in-patients in 176 hospitals with suspected or confirmed COVID-19.
First results could become available by the end of June, with further outcomes being delivered later in the summer.
'Modest Effects'
Speaking at a briefing on Thursday convened by the Science Media Centre, Prof Martin Landray, the trial's deputy chief investigator, said: "There's not likely to be any single one big winner. I think for all of us, that is extraordinarily unlikely, but much more likely that several drugs may have what we might think of as 'modest' effects.
"So, for example, if we could find a drug that lowered the risk of dying, which of course is the principal concern in this context, by just one fifth, that would turn out to be very important. One fifth doesn't sound like very much – it's not a cure – but if we found several drugs that each reduce the risk by one fifth, then quickly we'd be reducing the risk of dying by, say, a half or more."
Martin Landray, who is professor of medicine and epidemiology at the Nuffield Department of Population Health, pointed out that the use of multiple drugs for other diseases, such as diabetes, heart diseases, and HIV, was commonplace.
He said those involved in the RECOVERY trial did not yet know if any of the repurposed drugs would be successful in treating COVID-19 patients, but the large-scale nature of the trial could provide answers.
"The second thing to remember is that this is not a rare disease – this is a common disease," he said. "So to give you the UK context, sadly we're heading towards around 40,000 deaths in the UK. If one could reduce that by one fifth, then that would have been 8000 lives saved."
Drugs Being Trialled
The RECOVERY trial involves randomly assigning participants to receive either standard hospital care, or one of four active treatments.
The initial drugs being tested in the trial are:
- Lopinavir-ritonavir (commonly used to treat HIV)
- Low-dose dexamethasone (a type of steroid, which is used in a range of conditions typically to reduce inflammation)
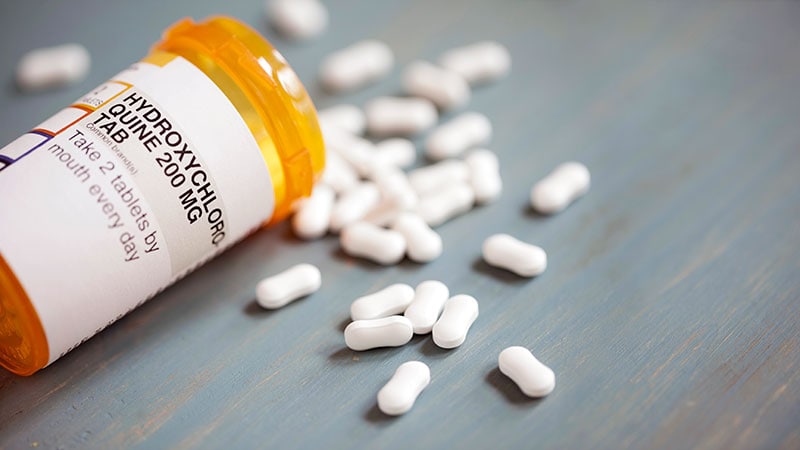
- Hydroxychloroquine (related to an anti-malarial drug)
- Azithromycin (a commonly used antibiotic)
In a second randomisation, patients with progressive COVID-19 receive either the immunosuppressive drug, tocilizumab (RoActemra, Roche), or standard hospital care.
Prof Peter Horby, the trial's chief investigator, said: "We are planning to add new drugs, as we move forward. We're going to keep the trial open.
"As we start to get answers on the drugs that we have, we will take them out of the trial. They'll either be ditched because they don't work or they'll become standard of care because they do, and then we can add in new drugs."
Peter Horby, who is professor of emerging infectious diseases and global health in the Nuffield Department of Medicine, said: "Currently, we're in the process of adding convalescent plasma as a new arm in the trial."
Convalescent plasma is collected from donors who have recovered from COVID-19 and contains antibodies against the SARS-CoV-2 virus.
Data on patient outcomes is being provided through NHS DigiTrials, which has made it possible for researchers to link trial data with NHS data sources.